- Blog
- Asus Eee Pc 900 Video Driver Windows 8
- Wiley Cpa Exam
- 8 Ball Of Crack Profit
- Lirik Lagu Sunda Orang Orangan Sawah
- Nokia 5130 Software Installation
- Cosmed Pft Software Suite
- Digital Photo Frame Key Ring Software Download
- Sara Zamana Haseeno Ka Deewana Free Mp3 Download
- Goldberg Music Mp3 Download
- Nasu Da Cartoon Episode 334 Full Video 3gp
- Driver Andromak U2
- Judul Judul Film Inazuma Eleven
- Download Lagu The Scientist Coldplay Original
- Carbylamine Reaction Pdf
- Kamichama Karin Sub Indo
- Download Game Bruce Lee Dragon Warrior Cho Android
- Sir James Jeans The Mysterious Universe Pdf Free
- Harry Potter Deathly Hallows Part 2 In Hindi Free Download
(i) Carbylamine reaction Carbylamine reaction is used as a test for the identification of primary amines. When aliphatic and aromatic primary amines are heated with chloroform and ethanolic potassium hydroxide, carbylamines (or isocyanides) are formed. These carbylamines have very unpleasant odours. Secondary and tertiary amines do not respond to this test. For example, (ii) Diazotization Aromatic primary amines react with nitrous acid (prepared in situ from and a mineral acid such as ) at low temperatures to form diazonium salts. This conversion of aromatic primary amines into diazonium salts is known as diazotization.
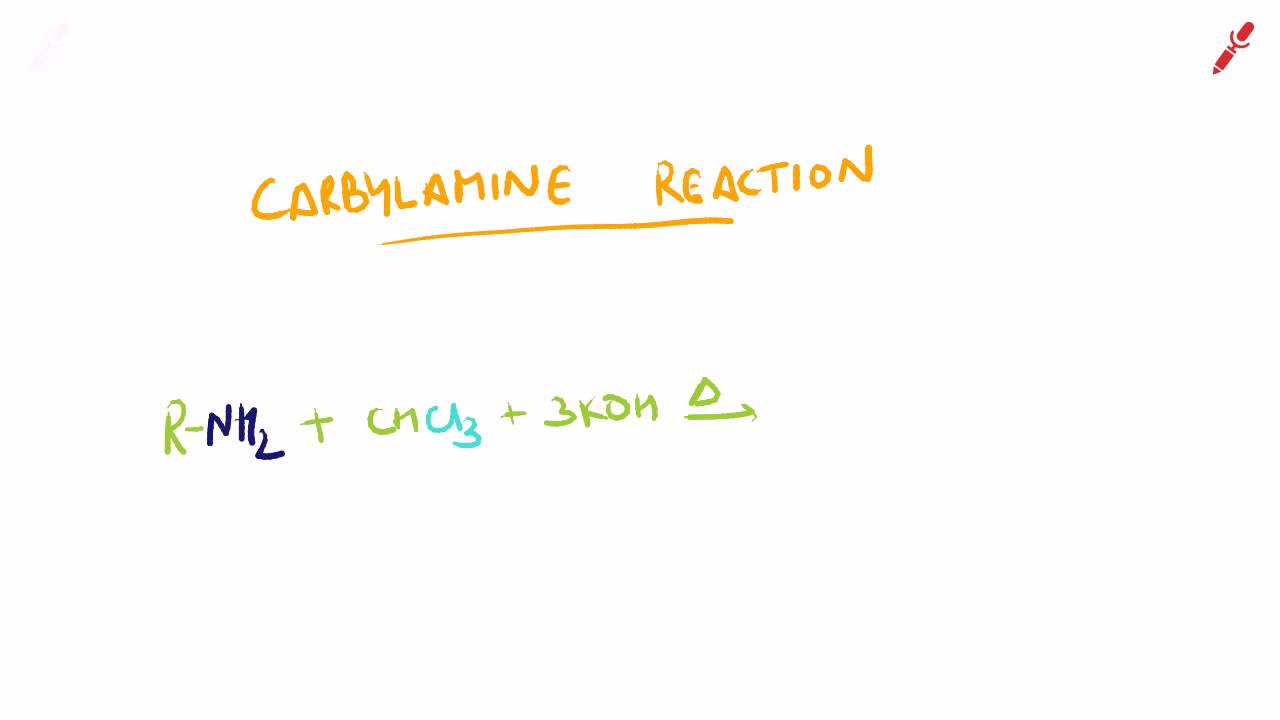
Organic Chemistry – Specific Name Reactions Class XII Sandmeyer Reaction The Cl. Carbylamine reaction. Carbylamine Reaction - Download as PDF File (.pdf), Text File (.txt) or read online. Carbylamine Reaction.
For example, on treatment with and HCl at, aniline produces benzenediazonium chloride, with and as by-products. (iii) Hofmann’s bromamide reaction When an amide is treated with bromine in an aqueous or ethanolic solution of sodium hydroxide, a primary amine with one carbon atom less than the original amide is produced.
This degradation reaction is known as Hoffmann bromamide reaction. This reaction involves the migration of an alkyl or aryl group from the carbonyl carbon atom of the amide to the nitrogen atom. For example, (iv) Coupling reaction The reaction of joining two aromatic rings through the bond is known as coupling reaction. Arenediazonium salts such as benzene diazonium salts react with phenol or aromatic amines to form coloured azo compounds.
Q 7 Iv 2 Coupling Reaction Q 7 iv 2 Coupling Reaction It can be observed that, the para-positions of phenol and aniline are coupled with the diazonium salt. This reaction proceeds through electrophilic substitution. (v) Ammonolysis When an alkyl or benzyl halide is allowed to react with an ethanolic solution of ammonia, it undergoes nucleophilic substitution reaction in which the halogen atom is replaced by an amino group. This process of cleavage of the carbon-halogen bond is known as ammonolysis. Q 7 Vi Acetylation Q 7 vi Acetylation Aliphatic and aromatic primary and secondary amines undergo acetylation reaction by nucleophilic substitution when treated with acid chlorides, anhydrides or esters. This reaction involves the replacement of the hydrogen atom of or group by the acetyl group, which in turn leads to the production of amides. To shift the equilibrium to the right hand side, the formed during the reaction is removed as soon as it is formed.
This reaction is carried out in the presence of a base (such as pyridine) which is stronger than the amine.
USE CODE: BCK2SKOOL Chemical Properties of Amines Table of Content. Basic Nature of Amines Like ammonia, amines are converted into their salts by aqueous mineral acids and are liberated from their salts by aqueous hydroxides.
Thus, like ammonia, amines are more basic than water and less basic than hydroxide ions. Amines turn red litmus blue and also combine with water and mineral acids to form corresponding salts. When the amine salts are treated with strong bases like NaOH, the parent amines are regenerated. RN + H 3 Cl – + OH – → RNH 2 + H 2 O + Cl – Amine salt Amine (Soluble is water) (Insoluble in water) Further, due to basic character amines react with auric and platinic chlorides in presence of HCl to form double salts. RNH 2 +PtCl 4 +2HCl - (RNH 3 ) 2 +PtCl 6 2- Chloroplatinic acid These double salts decompose on ignition to pure metal, therefore, the formation and decomposition of the double salts is used for determining the molecular weight of amines.
Alkylation of Amines?Like ammonia, an amine can react with alkyl halide to form next higher class of amine. Here, again it is the presence of electron pair on nitrogen which makes amine to behave as nuclephile and alkyl halide thus undergo nucleophilic substitution reaction. RNH 2 RNHCH 3 RN(CH 3) 2 RN +(CH 3) 3I 1 0 amine 2 0 amine 3 0 amine Quaternary ammonium iodide Quarternary ammonium salts are useful in synthetic organic chemistry as phase-transfer catalysts and in the preparation of alkenes1 Halogenation of Amines In order to introduce only one halogen atom, the activating effect of the –NH 2 group must be lowered using acetylation.
Nitration of Amines Direct nitration of aniline with nitric acid gives a complex mixture of mono – di and trinitro compounds and oxidation products. If however, NH 2 group is protected by acetylation, main product of nitration is p – nitro derivative. Sulphonation of Amines Aniline reacts with conc. H 2SO 4 to form the salt anilinium hydrogen sulphate which on heating at 455 – 475 K gives sulphanilic acid (p – amino benzene sulphonic acid). Sulphanilic acid exists as Zwitter ion i.e.
A dipolar ion which exists in the form of internal salt structure. Such ion has positive as well as negative charge within same molecular structure. Acylation of Amines Reaction refers tro the reaction of amines with acyl chlorides or acid anhydrides. Primary and secondary amines can react with acid chlorides or acid anhydrides to form substituted amides. RNH 2 + R’COCl → R’CO NHR an N-substituted amide R 2NH + R’COCl → R’CO.NR 2 an N,N disubstituted amide. Benzoylation of Amines (Schotten Baumann Reaction) Primary amine reacts with benzoyl chloride to give the acylated product.
